
If solvents are used to dissolve solids, care must be taken to avoid obscuring important spectral regions by solvent absorption. Liquids are usually examined as a thin film sandwiched between two polished salt plates (note that glass absorbs infrared radiation, whereas NaCl is transparent). Infrared spectra may be obtained from samples in all phases (liquid, solid and gaseous). Simply enter the value to be converted in the appropriate box, press " Calculate" and the equivalent number will appear in the empty box. A calculator for interconverting these frequency and wavelength values is provided on the right. Most infrared spectra are displayed on a linear frequency scale, as shown here, but in some older texts a linear wavelength scale is used. Wavelength units are in micrometers, microns (μ), instead of nanometers for the same reason. The reciprocal centimeter is the number of wave cycles in one centimeter whereas, frequency in cycles per second or Hz is equal to the number of wave cycles in 3*10 10 cm (the distance covered by light in one second). The frequency scale at the bottom of the chart is given in units of reciprocal centimeters (cm -1) rather than Hz, because the numbers are more manageable. Thus a sample that did not absorb at all would record a horizontal line at 100% transmittance (top of the chart). The inverted display of absorption, compared with UV-Visible spectra, is characteristic. Further analysis (below) will show that this spectrum also indicates the presence of an aldehyde function, a phenolic hydroxyl and a substituted benzene ring. The gap in the spectrum between 700 & 800 cm -1 is due to solvent (CCl 4) absorption. The complexity of this spectrum is typical of most infrared spectra, and illustrates their use in identifying substances. An example of such a spectrum is that of the flavoring agent vanillin, shown below. Infrared spectrometers, similar in principle to the UV-Visible spectrometer described elsewhere, permit chemists to obtain absorption spectra of compounds that are a unique reflection of their molecular structure. Consequently, virtually all organic compounds will absorb infrared radiation that corresponds in energy to these vibrations. We must now recognize that, in addition to the facile rotation of groups about single bonds, molecules experience a wide variety of vibrational motions, characteristic of their component atoms. The mobile nature of organic molecules was noted in the chapter concerning conformational isomers. The covalent bonds in molecules are not rigid sticks or rods, such as found in molecular model kits, but are more like stiff springs that can be stretched and bent. Photon energies associated with this part of the infrared (from 1 to 15 kcal/mole) are not large enough to excite electrons, but may induce vibrational excitation of covalently bonded atoms and groups. The portion of the infrared region most useful for analysis of organic compounds is not immediately adjacent to the visible spectrum, but is that having a wavelength range from 2,500 to 16,000 nm, with a corresponding frequency range from 1.9*10 13 to 1.2*10 14 Hz. On the immediate high energy side of the visible spectrum lies the ultraviolet, and on the low energy side is the infrared. This equation allows computing the number of degrees of freedom (F) from the number of components (C) and the number of phases in the system (P).As noted in a previous chapter, the light our eyes see is but a small part of a broad spectrum of electromagnetic radiation. If both are constant, change the value to "0". If one of these variables is constant, you have to change its value to "1". /Chi-square_distributionCDF-English-5941084d5f9b58d58a344569-5b8ff0cec9e77c005082389b.jpg)
factor (by default = 2) – An integer value in the equation that depends on the temperature and pressure of the system (see the pressure calculator).P – Number of phases in the system and. 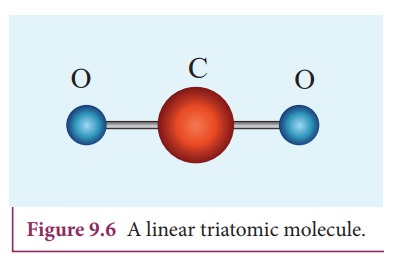
The phase rule equation proposed by Josiah Willard Gibbs in 1875 (American theoretical physicist, a professor at Yale University) is expressed by the formula: Moreover, the phase rule allows the creation of phase diagrams to represent and interpret phase equilibria in heterogeneous systems. It predicts the equilibrium relations of phases and is present as a function of physical conditions such as pressure and temperature. 
Gibbs' phase rule is based on the laws of thermodynamics and provides the theoretical principle for characterizing the chemical state of a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
